Elements, compounds, mixtures, solutions, environments ..
Sun and air?
Study the seaside image for a few seconds . Try to describe it:
You might say that the image shows : A calm sea, clear blue sky, bright sunshine and a perfectly smooth beach.
You could describe it in terms of the states of matter visible:
| SOLID | Sand |
| LIQUID | Sea |
| GAS | Air |
| GAS and PLASMA | Sun |
As a chemistry student you might be asked to classify each of these four components as either mixtures, compounds or elements.
As a biology student you might be asked to describe the scene in terms of environments and ecosystems...
You might even use words like Atmosphere, Hydrosphere and Lithosphere ?
Some elements up close
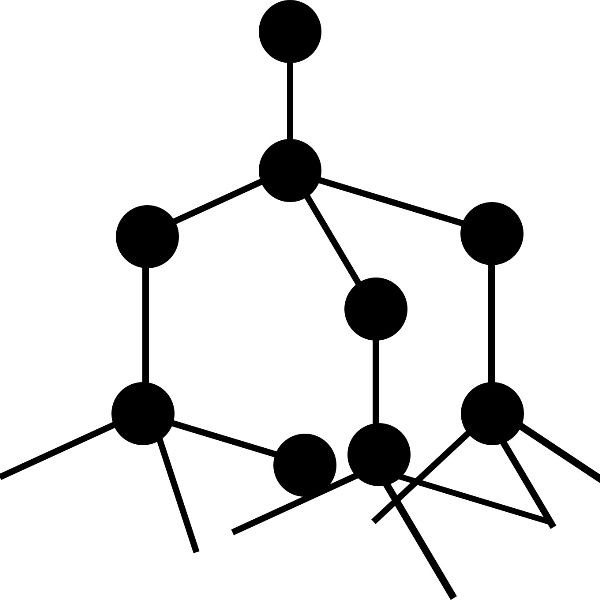
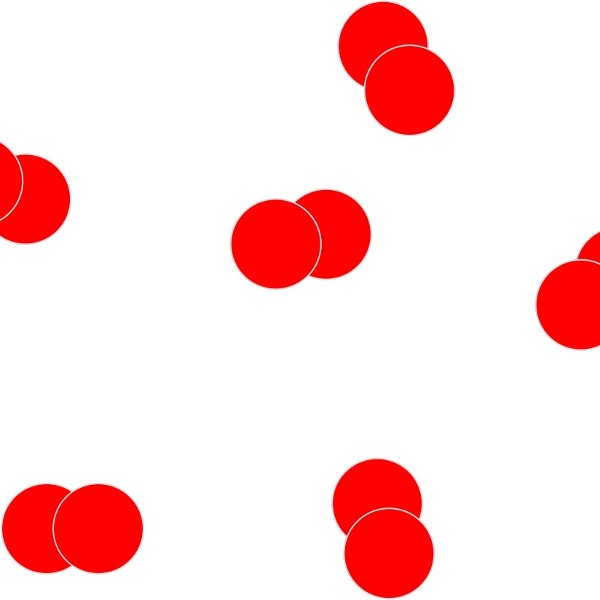
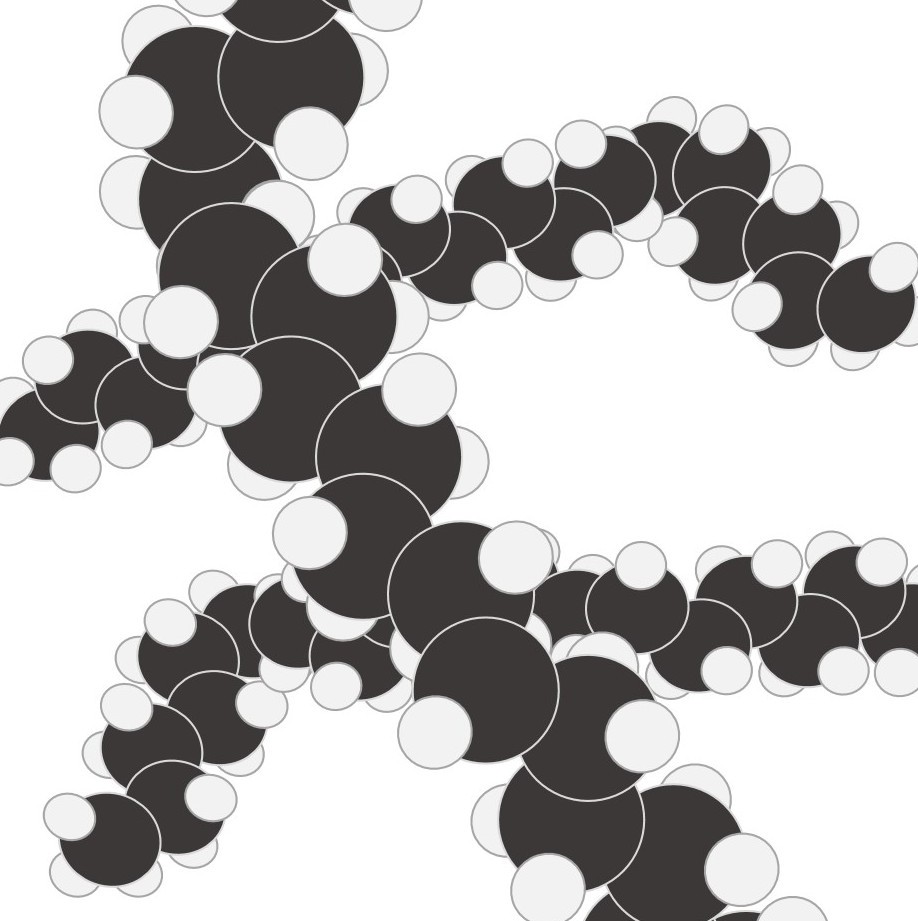
Simple molecular elements. These are often gases at room temperature but can be liquid or easily melted solids.
The atoms shown here are paired up to form diatomic molecules. The forces between the molecules are weak and so elements with this structure will have low melting points and might even be gases at room temperature.
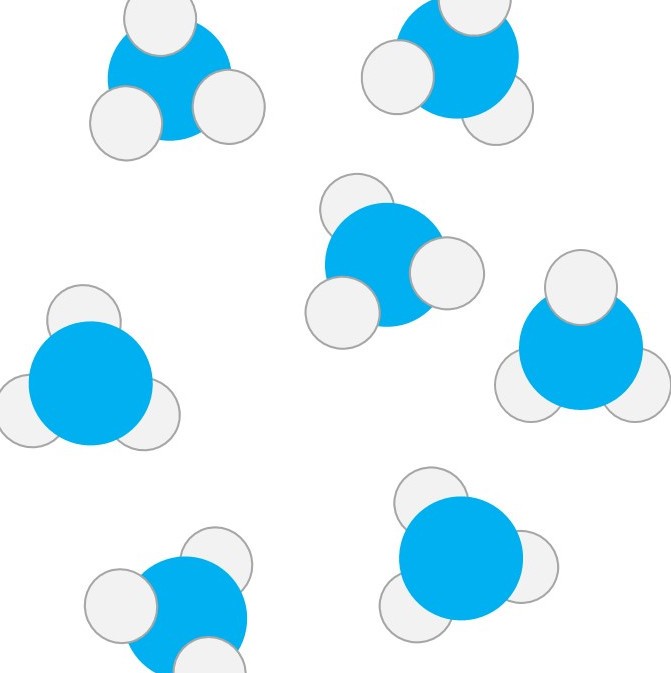
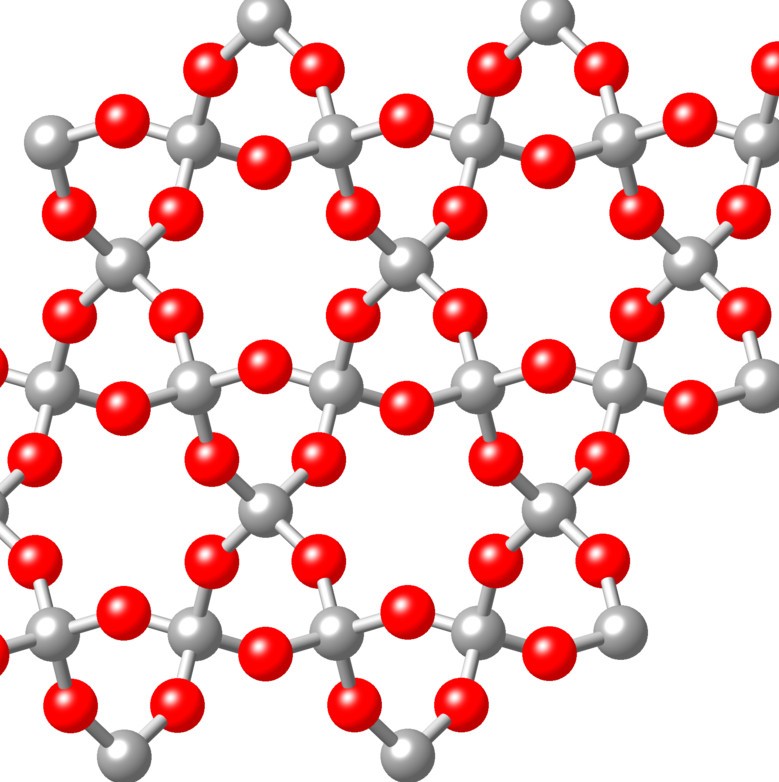
Some compounds up close
Activity. Complete the matching exercise below
When you subscribe to the blog, we will send you an e-mail when there are new updates on the site so you wouldn't miss them.